Early appearance of two distinct genomic lineages of SARS-CoV-2 in different Wuhan wildlife markets suggests SARS-CoV-2 has a natural origin
Robert F. Garry1,2
1Department of Microbiology and Immunology, Tulane University Medical Center, 1430 Tulane Avenue, New Orleans, Louisiana 70112 USA; E-Mail: [email protected]
2Zalgen Labs, LLC, Germantown, MD, USA
Introduction
Our previous commentary on the Proximal Origins of SARS-CoV-2 (Andersen et al., 2020) concluded that, “SARS-CoV-2 is not a laboratory construct or a purposefully manipulated virus." The possibility of a laboratory release or Lab Leak was also considered, but it was determined that a natural origin of SARS-CoV-2 is much more likely. Recently, a team of scientists under the auspices of the World Health Organization (WHO) reached similar conclusions (WHO, 2021). Other groups have speculated that SARS-CoV-2 may be the result of undisclosed research on SARS-CoV-2 or a close progenitor and accidental release (Relman, 2020; Butler et al., 2021). It has also been suggested that SARS-CoV-2 may be the product of laboratory passage of a progenitor virus in cell culture or animals or Gain-of-Function research (Relman, 2020; Segreto and Deigin, 2020; Sirotkin and Sirotkin, 2020, Yan et al., 2020; Zhan et al., 2020). New data presented by the WHO study provides clear findings in support of the natural origin of SARS-CoV-2.
Results
The study of SARS-CoV-2 origins conducted by the WHO team provided important new data regarding the role of wildlife markets in the emergence of SARS-CoV-2. The large Huanan seafood market, which also sold wildlife and wildlife products, was a focus of attention because it was linked to the majority of early cases of COVID-19 in Wuhan. The WHO report documented that early cases were not only linked to the Huanan market, but that other early cases were linked to different markets that sold wildlife or wildlife products. Among the first 168 diagnosed cases of COVID-19 in Wuhan with onset date prior to December 31, 2019 and a known history of exposure to wildlife markets 55.4% (93/168) reported such exposures. Of the 168 cases, 28% (47/168) had only been to the Huanan market, 22% (38/168) had exposure to another wildlife market and 4.7% (8/168) had exposure to the Huanan market and another market (Annex E2, Table 1 of WHO, 2021).
The genetic lineages of SARS-CoV-2 associated with early cases in Wuhan were documented in the WHO report. Previously, Rambaut et al, (2020) noted that at the root of the phylogeny of SARS-CoV-2 are two lineages designated lineage A and B. Early lineage A viruses include SARS-Cov-2 isolate EPI_ISL_529213 sampled on 30Dec19 from a person linked to a wildlife market different than the Huanan market (Molecular Epidemiology Table 7, sample 13 in WHO, 2021). Linage A viruses share two nucleotides (T8,782 in ORF1ab and C28,144 in ORF8) with the bat viruses RaTG13 and RmYN02 and other sarbecoviruses. It is likely that the most recent common ancestor (MRCA) of SARS-CoV-2 shares the same genome sequence as these early lineage A sequences (Rambaut et al., 2020). Different nucleotides (C8,782 in ORF1ab and T28,144 in ORF8) are present at those sites in viruses assigned to lineage B, such as SARS-CoV-2 isolate Wuhan-Hu-1 (GenBank accession no. MN908947, Molecular Epidemiology Table 7, sample 06 in WHO, 2021) sampled from the Huanan market on 30Dec19. All virus positive samples from the Huanan market, from venders or customers of the market or from environmental samples, contained SARS-CoV-2 of lineage B. There was limited genetic diversity in the lineage B samples from the Huanan market, which is consistent with the market as a site of a super-spreader event. Lineage A and Lineage B viruses spread throughout Wuhan and to other countries (Rambaut et al., 2020; Worobey et al., 2020).
Competing hypotheses have been put forward to explain the emergence of SARS-CoV-2. Natural spillover directly from a bat or via an intermediate animal host could have occurred via several different scenarios (Fig. 1). Likewise, several variations of the Lab Leak hypothesis have been proposed (Fig. 2). A dispassionate science-based discourse on the topic of the origin of SARS-CoV-2 must account for this new data revealed by the WHO study showing: 1. multiple markets were linked to the early cases, and 2. divergence of SARS-CoV-2into lineages A and B was an early occurrence. These facts are represented by yellow boxes in each figure.
Figure 1. Natural scenarios of SARS-CoV-2 origin.
Natural spillover directly from a bat or via an intermediate host may have involved trade in wildlife susceptible to SARS-CoV-2, either trapping and hunting of wildlife in nature or farming of “wildlife species.” There is now clear evidence that SARS-CoV-2 is capable of effective spread, not only in humans, but to a diverse group of mammals (Garry, 2020). The multi-market aspect of the early outbreak can be explained by distribution of SARS-CoV-2 infected animals to more than one market (Fig. 1). In Natural scenarios diversification of SARS-CoV-2 to lineages A and B could have occurred prior to the distribution, either at a wildlife farm or during transport of the animals to the markets. It is possible that humans involved in the wildlife trade were also infected and involved in this pathway.
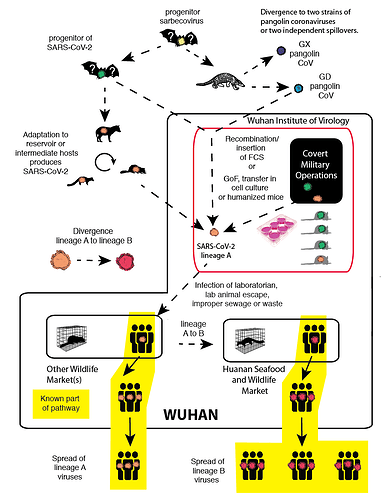
Figure 2. Lab Leak scenarios of SARS-CoV-2 origin.
Proponents of Lab Leak theories speculate that SARS-CoV-2 or a close progenitor was present in the Wuhan Institute of Virology or another Wuhan virology lab before the start of the COVID-19 pandemic and the appearance of the first cases in Wuhan (Fig. 2). It has been proposed that SARS-CoV-2 was released either via an infected laboratory worker, an escaped lab animal or via waste disposal (Relman 2020; Butler et al., 2021). Pangolins carry and appear to be naturally infected with at least two lineages of sarbecoviruses (Liu et al., 2019; Lam et al., 2020). The Guangdong strain of pangolin coronavirus carries a receptor binding domain (RBD) that is highly similar to the RBD of SARS-CoV-2 (Lam et al., 2020, Andersen et al., 2020). Some proponents of the Lab Leak theory have speculated the RBD of GD pangolin was recombined via genetic engineering with the backbone of an undisclosed sarbecovirus to produce SARS-CoV-2. Alternatively, it has been suggested that an undisclosed close progenitor of SARS-CoV-2 was passaged on human cells or experimental animals (such a humanized mice) in gain-of-function type experiments to adapt for human replication (Relman, 2020, Segreto and Deigin, 2020; Sirotkin and Sirotkin, 2020). The direct role of the Chinese military in conducting classified research on sarbecoviruses at the WIV has also been proposed (United States, 2021). In Lab Leak scenarios diversification of SARS-CoV-2 to its two separate early lineages A and B would have had to occur in the laboratory setting. Lab Leak scenarios must also account for the fact that the majority of early cases were associated with different wildlife markets in Wuhan.
Discussion
Lab Leak scenarios are inconsistent with several established facts regarding the origin of SARS-CoV-2. The majority of early cases were linked to different markets that sold wildlife or wildlife products in Wuhan. All theories of the origin of SARS-CoV-2 must account for the linkage to different market engaged in wildlife trade. Theories on SARS-CoV-2 must also account for the fact that two distinct lineages of SARS-CoV-2 were distributed at different Wuhan wildlife markets. Scenarios where an infected laboratory worker, an escaped lab animal or faulty waste disposal spread not one but two lineages of SARS-CoV-2 specifically to different wildlife markets are difficult to rationalize.
The original SARS-CoV outbreaks in 2002-2004 were linked to the wildlife trade (Guan et al., 2003). In contrast to Lab Leak theories, linkage of the origin of SARS-CoV-2 to wildlife or the wildlife trade provides several plausible scenarios for the appearance of SARS-CoV-2 at different wildlife markets. It fully accounts for the fact that the majority of early Wuhan COVID-19 cases were linked to different wildlife markets in a straightforward manner. It also provides simple explanations for the fact that two different lineages of SARS-CoV-2 were linked to markets. In one possible scenario divergence of SARS-CoV-2 to lineages A and B occurred prior to the transport of infected animals to Wuhan and the infected animals were subsequently distributed to different wildlife markets.
Proponents of the Lab Leak theory will point out that none of the animals sold at the Huanan Market tested positive for SARS-CoV-2 (WHO, 2021). Although mentioned in the WHO Report, but not discussed in detail, several independent sources indicate that wildlife species susceptible to SARS-CoV-2, including civets and raccoon dogs, were sold at the Huanan market (Stout, 2020; Yee, 2020; Zhang and Holmes, 2020). Similar species were likely to have been available to purchase at other wildlife markets in Wuhan. Certain species of animals may have been removed after the appearance of the first COVID-19 cases and the linkage of COVID-19 cases to the market, but prior to the closure of the Market on January 1, 2020. It should also be noted that environmental samples that did test positive were associated with the portion of the market where wildlife or wildlife products were sold. A temporal analysis of the early human cases at the Huanan market confirms the pattern of spread from the areas of the market where wildlife products were sold to other parts of the market. (WHO, 2021).
Hybrids of natural and Lab Leak scenarios have been suggested (Relman, 2020; Baker, 2020). Some variations on a hybrid scenario suggest that SARS-CoV-2 is a natural virus that infected a scientist while doing field work resulting in mildly symptomatic or asymptomatic spread or that after being brought back to a laboratory SARS-CoV-2 was released unknowingly, but not successfully cultured or otherwise manipulated. Compared to the millions of worldwide encounters of humans with wildlife, including the trapping of bats for food, the number of high-risk exposures of scientists doing field or laboratory work with samples from wildlife is miniscule. There are no documented cases of laboratory infections with previously unknown, but pathogenic, viruses. Thus, hybrid scenarios are of very low probability. They also fail to explain how two lineages of SARS-CoV-2 came to be distributed at the different wildlife markets.
Conclusions
New data compiled by the WHO team regarding the presence of distinct lineages of SARS-CoV-2 in different Wuhan wildlife markets are inconsistent with a laboratory-based origin of SARS-CoV-2. No data or other evidence has emerged in support of the Lab Leak theory. In contrast, the WHO report significantly adds to the large volume of epidemiological and genomic data that support emergence of SARS-CoV-2 from a zoonotic reservoir, either wildlife or farmed animals.
Acknowledgments
The important work of the WHO-convened Global Study of Origins of SARS-CoV-2 team is gratefully acknowledged. Kristian G. Andersen, Edward C. Holmes, Andrew Rambaut and William R. Gallaher provided essential input and discussion. Work on emerging viruses in the Garry Laboratory is supported by the National Institutes of Health, the Coalition for Epidemic Preparedness Innovations, the Burroughs Wellcome Fund, the Wellcome Trust, the Center for Disease Prevention and Control, and the European & Developing Countries Clinical Trials Partnership.
References
Andersen KG, Rambaut A, Lipkin WI, Holmes EC and Garry RF (2020). The proximal origin of SARS-CoV-2. Nat Med 26:450-452.
Baker N. (2021). The Lab-Leak hypothesis. Did COVID-19 Escape From a Lab? A Coronavirus Investigation
Butler CD, Canard B, Cap H, Chan YA, Jean-Michel Claverie J-M, Colombo F, Courtier V, de Ribera FA, Decroly E, Maistre R, Demaneuf G, Ebright RH, Goffinet A, Graner F, Halloy J, Leitenberg M, Lentzos F, McFarlane R, Metzl J, Petrovsky N, Quay S, Rahalkar MC, Segreto R, Theißen G, van Helden J. (2021). Call for a full and unrestricted international forensic investigation into the origins of COVID-19. Investigation into Covid Origins Sought - The New York Times
Garry RF (2020). https://pando.tools/t/mutations-arising-in-sars-cov-2-spike-on-sustained-human-to-human-transmission-and-human-to-animal-passage/578
Guan Y, Zheng BJ, He YQ, Liu XL, Zhuang ZX, Cheung CL, Luo SW, Li PH, Zhang LJ, Guan YJ, Butt KM, Wong KL, Chan KW, Lim W, Shortridge KF, Yuen KY, Peiris JS, Poon LL. (2003). Isolation and characterization of viruses related to the SARS coronavirus from animals in southern China. Science 302:276-8.
Lam TT, Jia N, Zhang YW, Shum MH, Jiang JF, Zhu HC, et al. (2020). Identifying SARS-CoV-2-related coronaviruses in Malayan pangolins. Nature 583:282-5.
Liu P, Chen W and Chen, JP. (2019). Viral metagenomics revealed Sendai virus and coronavirus infection of Malayan pangolins (Manis javanica). Viruses 11:979.
Rambaut A, Holmes EC, O’Toole et al. (2020). A dynamic nomenclature proposal for SARS-CoV-2 lineages to assist genomic epidemiology. Nat Microbiol 5:1403–1407.
Segreto R and Deigin Y. (2020). The genetic structure of SARS-CoV-2 does not rule out a laboratory origin: SARS-COV-2 chimeric structure and furin cleavage site might be the result of genetic manipulation. BioEssays : news and reviews in molecular, cellular and developmental biology, e2000240.
Sirotkin K. and Sirotkin D. (2020). Might SARS-CoV-2 have arisen via serial passage through an animal host or cell culture?: A potential explanation for much of the novel coronavirus’ distinctive genome. BioEssays : news and reviews in molecular, cellular and developmental biology 42, e2000091
Stout KL (2020). “Wuhan SARS”: Tracing the origin of the new virus to China’s wild animal markets. “Wuhan SARS”: Tracing the origin of the new virus to China’s wild animal markets - YouTube.
United States (Department of State). (2021). Fact Sheet: Activity at the Wuhan Institute of Virology. Fact Sheet: Activity at the Wuhan Institute of Virology - United States Department of State.
WHO, 2021. “WHO-convened global study of origins of SARS- CoV-2: China part”; www.who.int/publications/i/item/who-convened-global-study-of-origins-of-sars-cov-2-china-part.
Worobey M, Pekar J, Larsen BB, Nelson MI, Hill V, Joy JB, Rambaut A, Suchard MA, Wertheim JO, Lemey P. (2020). The emergence of SARS-CoV-2 in Europe and North America. Science 370:564-570.
Yan L-M, Kang S, Guan J and Hu S. (2020). Unusual features of the SARS-CoV-2 genome suggesting sophisticated laboratory modification rather than natural evolution and delineation of its probable synthetic route. Unusual Features of the SARS-CoV-2 Genome Suggesting Sophisticated Laboratory Modification Rather Than Natural Evolution and Delineation of Its Probable Synthetic Route | Zenodo.
Yee J. (2020). Bizarre Wuhan wet market menu shows over 100 wild animals sold as food.
Bizarre Wuhan Wet Market Menu Shows Over 100 Wild Animals Sold As Food, Link With Virus Unclear.
Zhan S, Deverman B, and Chan Y. (2020). SARS-CoV-2 is well adapted for humans. What does this mean for re-emergence? SARS-CoV-2 is well adapted for humans. What does this mean for re-emergence? | bioRxiv.
Zhang YZ and Holmes EC. (2020). A Genomic Perspective on the Origin and Emergence of SARS-CoV-2. Cell 181:223-227.